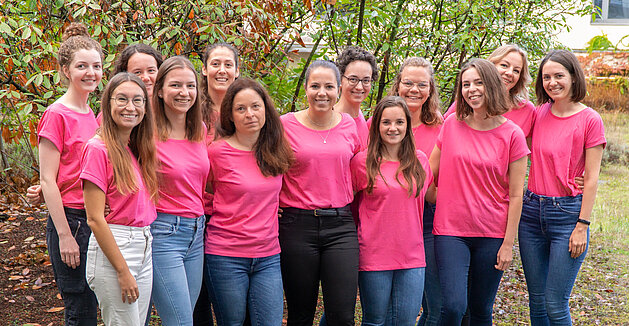
We Go Pink for Breast Cancer Awareness
October 1 marks the beginning of Breast Cancer Awareness Month, and the team of Mint Ladies from our Heidelberg office is wearing pink today.
We wear…
Read more