
From PCWG3 to PCWG4: Evolving Standards in Prostate Cancer Clinical Trials
The Prostate Cancer Working Group 4 (PCWG4) updates and extends the recommendations of PCWG3 to reflect a patient-centric approach, the emergence of…

The Prostate Cancer Working Group 4 (PCWG4) updates and extends the recommendations of PCWG3 to reflect a patient-centric approach, the emergence of…

Requests for radiological clinical trial evaluations frequently rely on fragmented methods such as emails, phone calls, and spreadsheets. This…
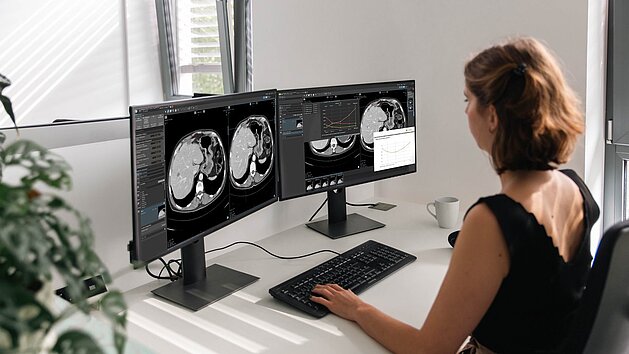
For decades, oncology trials have relied on RECIST 1.1 to evaluate drug efficacy. However, these categorical "snapshot" evaluations do not always…
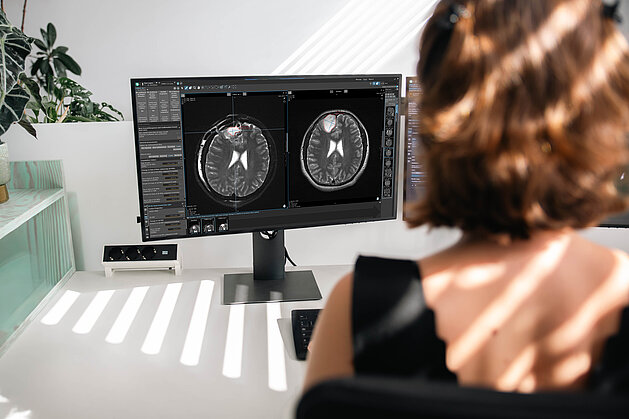
Tumor response assessment in neuro-oncology clinical trials requires careful attention to measurement protocols and confirmation scan requirements. To…

At Mint Medical, we know that organized and high-quality data is the foundation of successful research. That's why we've developed the Template…

The ESOI-EORTC workshop, hosted by the European Society of Oncological Imaging (ESOI) and the European Organization for Research and Treatment of…

Interoperability is revolutionizing healthcare by enabling the seamless exchange of patient data across systems. This efficient data flow is critical…
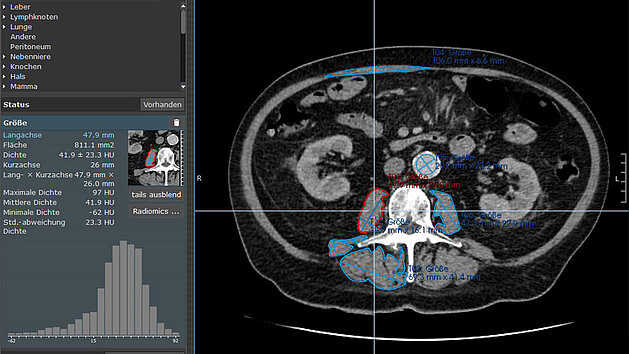
A recent study [1] conducted by researchers at University Hospital Ulm explored the role of sarcopenia in patients with esophageal or gastric cancer.…
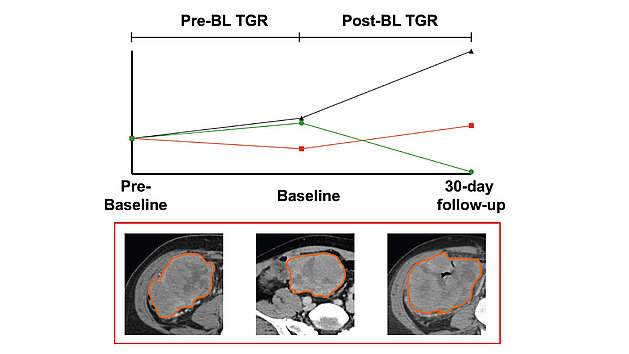
A recent study[1] conducted by researchers at the University Hospital Munich (LMU) investigated the role of tumor growth rate (TGR) in predicting the…