
Categories:




NCCN 2026: Memorial Sloan Kettering Cancer Center Presents Research on Structured Tumor Response Assessments with mint Lesion
Big congratulations to Steven Philemond and Alison Chiaramonte of Memorial Sloan Kettering Cancer Center for presenting their research poster “Use of…
Read more

Supporting Pediatric Brain Tumor Research with Structured Imaging Data
Brain tumors account for more than 15% of childhood cancers and are the second most common childhood cancer [1]. Despite significant advances in…
Read more


RÖKO 2026 – A Look Back at Innovation, Collaboration and Lung Cancer Screening
The 107th German radiology congress (RÖKO) in Leipzig brought together inspiring conversations, new ideas around AI and structured reporting, and…
Read more
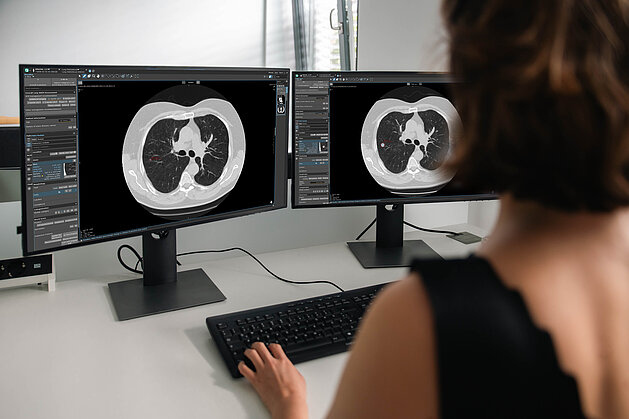
How Radiology Practices Can Participate in Germany’s Lung Cancer Screening Program with mint Lesion
New opportunities - but also economic uncertainty
With the launch of lung cancer screening (LCS) in Germany, radiology practices are facing a new…
Read more