A recent retrospective study led by Dr. Marilyn J. Siegel and her team at the Washington University School of Medicine in St. Louis has shed light on a critical issue in cancer care: routine clinical reads are more prone to overdiagnosing progressive disease when compared to RECIST 1.1 interpretations. This discrepancy holds significant implications, potentially leading to the premature discontinuation of effective treatments for cancer clinical trial participants and patients under standard care.
In this study, mint Lesion software was utilized for the criteria-based reads, determining overall response assessments according to RECIST 1.1 criteria, and generating structured reports for the clinical trial's principal investigator.
To learn more about the study's insights into the discrepant assessments and the suggested steps for mitigating this issue, click here.
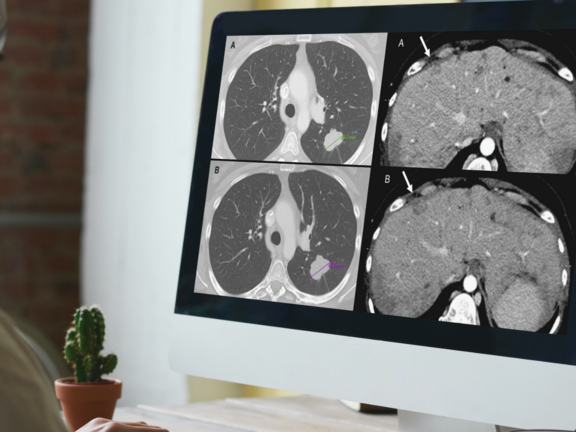
Study Discovers Overdiagnosis of Progressive Cancer in Routine Clinical Evaluations
Related Resources
Related Resources
Structured Reporting in Radiology: Precision, Clarity, and Data Quality
Radiology reports are often the key to a diagnosis – and the way they are created and communicated makes a crucial difference. Traditional free-text…

Lung Cancer Screening in Germany – A Turning Point for Early Detection
Few topics are currently as impactful in radiology as lung cancer screening. From 2026, Germany is expected to roll out a nationwide program, marking…

Standardizing Oncological Imaging: The BORN Project
The BZKF BORN Project (Bavarian Oncological Radiology Network) is setting new standards in the collection and evaluation of oncological imaging data.…