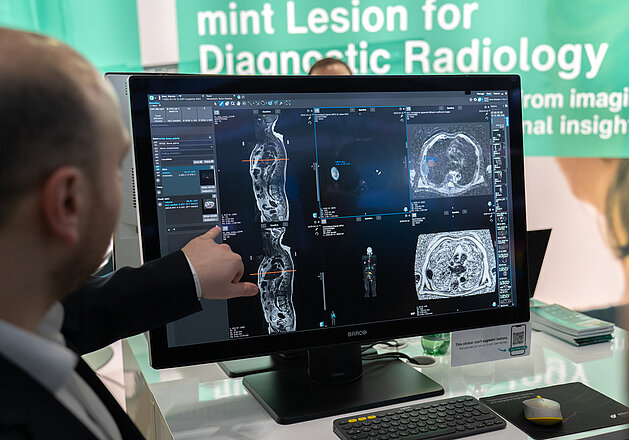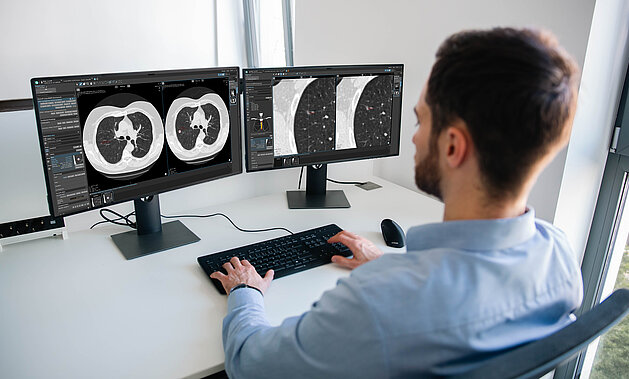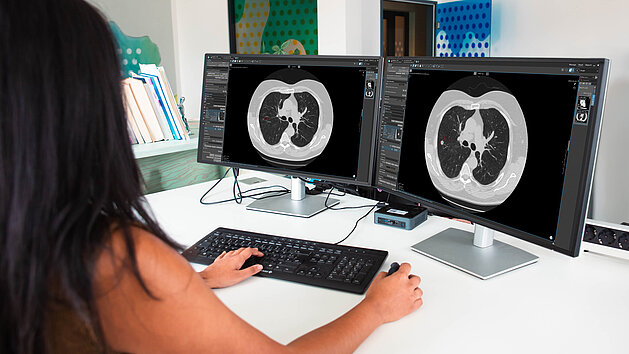
Lung Cancer Screening in Germany: How mint Lesion Supports Hospitals with Infrastructure, Integration, and Scalability
Screening as a Strategic Challenge
With the launch of the national lung cancer screening program in 2026, hospitals and screening centers across…
Read more