A recent retrospective study led by Dr. Marilyn J. Siegel and her team at the Washington University School of Medicine in St. Louis has shed light on a critical issue in cancer care: routine clinical reads are more prone to overdiagnosing progressive disease when compared to RECIST 1.1 interpretations. This discrepancy holds significant implications, potentially leading to the premature discontinuation of effective treatments for cancer clinical trial participants and patients under standard care.
In this study, mint Lesion software was utilized for the criteria-based reads, determining overall response assessments according to RECIST 1.1 criteria, and generating structured reports for the clinical trial's principal investigator.
To learn more about the study's insights into the discrepant assessments and the suggested steps for mitigating this issue, click here.
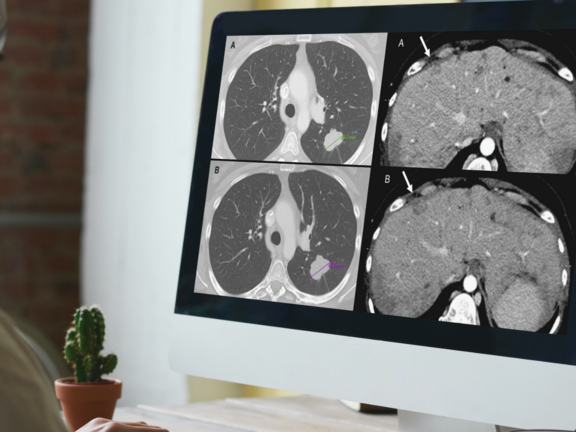
Study Discovers Overdiagnosis of Progressive Cancer in Routine Clinical Evaluations
Related Resources
Related Resources
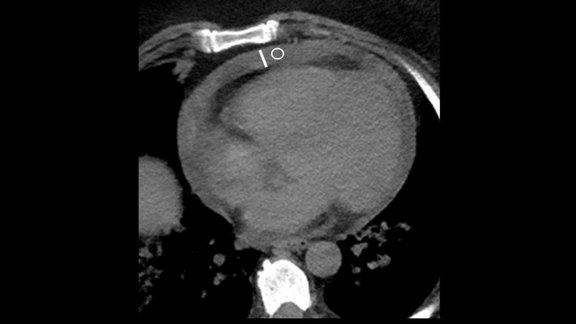
RACOON: Study emphasizes the clinical relevance of pericardial effusion as an imaging biomarker in COVID-19 patients
A multicenter study [1] based on the German research infrastructure project RACOON (Radiological Cooperative Network of the COVID-19 pandemic; a Netzw…

Software-Assisted CT Assessment Outperforms Manual Methods in Oncology Study
A recent study conducted at UKE Hamburg compared manual and software-assisted assessments of computed tomography (CT) scans according to iRECIST…
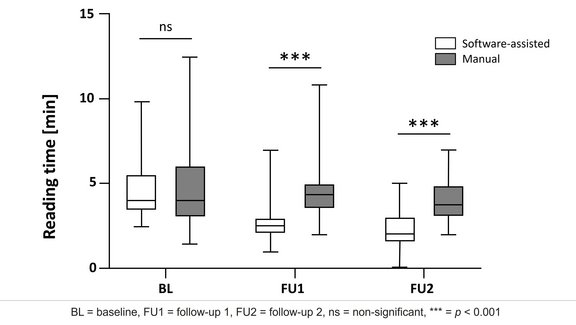
UKE Hamburg: Study Shows that Software-Assisted Assessments Enhance iRECIST Evaluation
This research study [1] aimed to compare the feasibility and reliability of manual versus software-assisted assessments of computed tomography (CT)…