
From PCWG3 to PCWG4: Evolving Standards in Prostate Cancer Clinical Trials
The Prostate Cancer Working Group 4 (PCWG4) updates and extends the recommendations of PCWG3 to reflect a patient-centric approach, the emergence of…

The Prostate Cancer Working Group 4 (PCWG4) updates and extends the recommendations of PCWG3 to reflect a patient-centric approach, the emergence of…

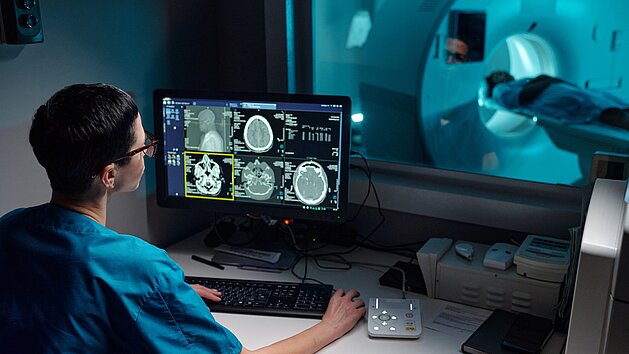
Postoperative MRI after brain tumor surgery is often affected by artifacts caused by intracranial air, limiting reliable image interpretation.
A…

Requests for radiological clinical trial evaluations frequently rely on fragmented methods such as emails, phone calls, and spreadsheets. This…
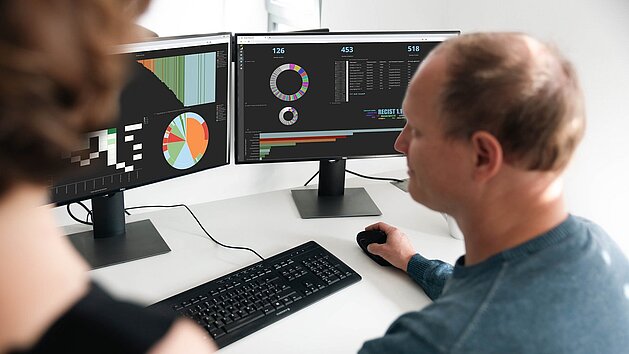
For many investigators, the transition from a single-site study to a multicentric model…
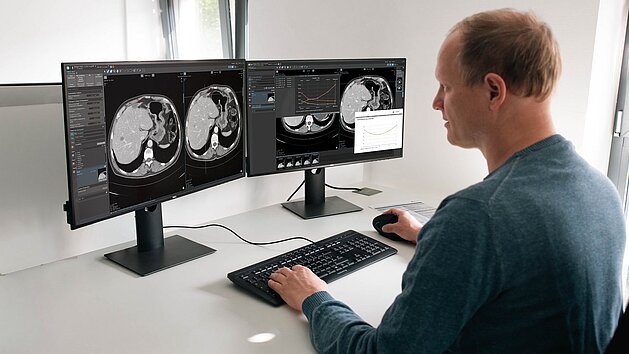
Following the FDA’s recent analysis of the growth rate constant (g) as a prognostic marker1, the challenge for many research organizations is…
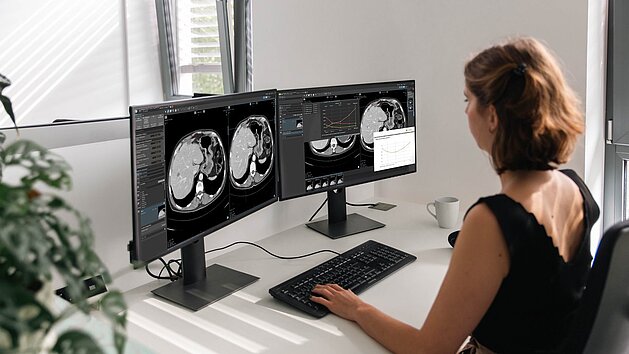
For decades, oncology trials have relied on RECIST 1.1 to evaluate drug efficacy. However, these categorical "snapshot" evaluations do not always…
How can complex tumor findings be assessed accurately, reproducibly, and in line with clinical guidelines?