The prospective study conducted by LMU Klinikum Munich investigates the treatment of localized prostate cancer using two techniques: vascular-targeted photodynamic therapy (VTP) and high-intensity focused ultrasound (HIFU).
The aim of the study is to use multiparametric magnetic resonance imaging (mpMRI) to track the anatomical and functional changes in the prostate and tumor post-treatment, as well as to detect possible recurrences.
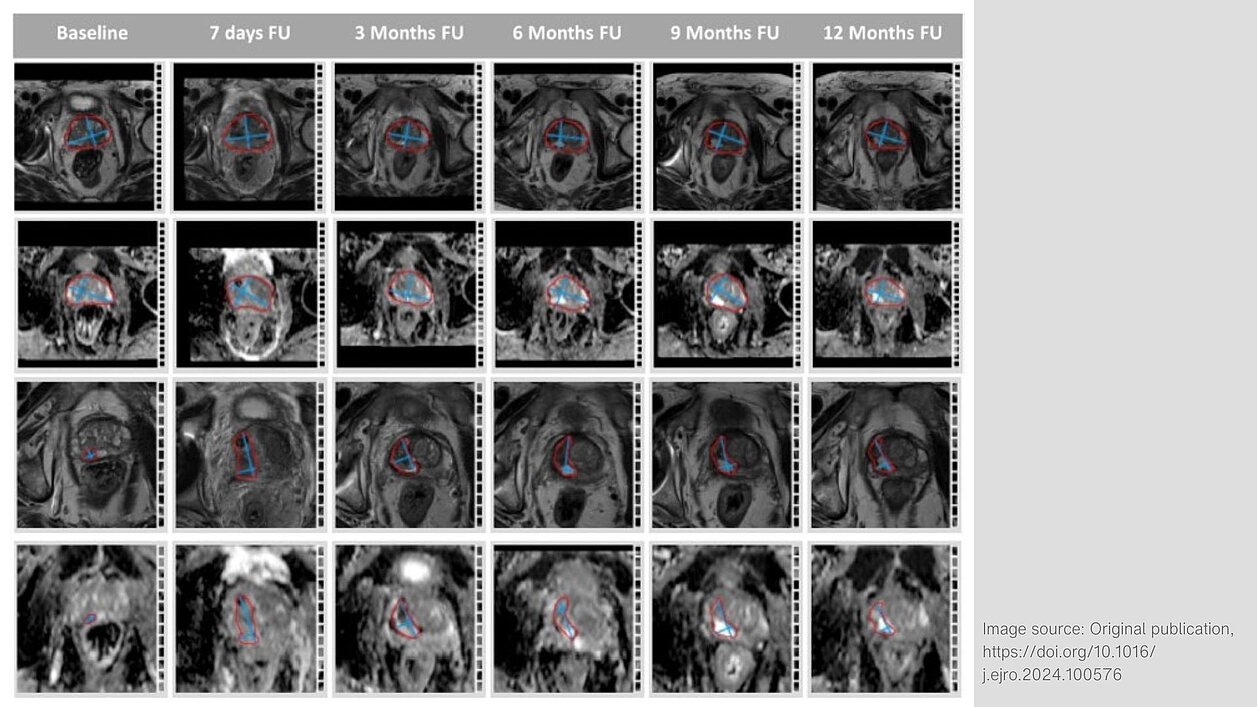
The study examines changes in mpMRI scans following focal therapies (FT), specifically VTP and HIFU, to monitor tumor response and prostate changes. mpMRI is a valuable tool for non-invasive monitoring, though it poses challenges in terms of standardization.
For the study, ten patients with localized prostate cancer (PCa) were treated with either VTP (70%) or HIFU (30%) between December 2020 and September 2021. They were followed up with mpMRI scans at several intervals (7 days and 3, 6, 9, and 12 months after treatment). The patients had an average age of 66.6 years (range: 58-80).
The researchers analyzed changes in prostate gland volume (PGV) and tumor volume (TV), as well as signal intensities in the prostate and tumor areas, using the software mint Lesion™.
“To the best of our knowledge, no published prospective studies have reported mpMRI imaging findings of the prostate after FT using a dedicated semi-automatic software for image assessment. Most other studies evaluate mpMRI images using two independent readers.”
Both treatments led to an initial increase in prostate volume due to swelling after therapy, followed by a significant reduction over time.
“We found that changes of PGV and TV were most obvious 7 days after the treatment with significant increases of the size of the prostate and the former tumor volume. One year after FT the PGV decreased by 18.6% compared to the baseline and was significantly lower than the PSV [sic] at 7 days after the treatment. Our findings are consistent with other studies that have evaluated mpMRI after focal therapy.”
The tumor volume significantly decreased in most patients one year after treatment. The mpMRI scans showed changes such as necrosis (dead tissue) and scar formation in the ablation zone. In one patient, a tumor recurrence was detected on rebiopsy, which was identified as a suspicious lesion with diffusion restriction through mpMRI.
mpMRI can effectively monitor post-treatment changes in prostate cancer patients treated with FT. It shows potential for detecting tumor recurrence but requires further validation through larger studies to standardize postoperative imaging evaluation.
Read the original publication here: link.springer.com/article/10.1007/s00345-024-05143-6
Olga Solyanik, Maria Apfelbeck, et al. 2024. “Prospective close monitoring of the effect of vascular-targeted photodynamic therapy and high intensity focused ultrasound of localized prostate cancer by multiparametric magnetic resonance imaging.” World Journal of Urology 42.462.