Radiologists utilize Whole-Body MRI (WB-MRI) as an established imaging method for multiple myeloma staging [1,3]. Because it avoids ionizing radiation, WB-MRI provides a safe option for serial scans [1]. Furthermore, multi-parametric MRI sequences allow for the visualization of active disease, giving radiologists a clear avenue for quantitative disease burden assessment [1, 4].
However, when integrating comprehensive WB-MRI analysis into standard clinical workflows, radiologists encounter several structural challenges:
- Detecting and reporting disease across the entire skeleton is time-intensive [2].
- Quantifying total disease burden objectively is difficult within standard time constraints [4].
- Systematically assessing bone marrow heterogeneity presents methodological difficulties [2, 3].
- Qualitative, text-based reporting can introduce inter-reader variability [1].
AI Capabilities Integrated into mint Lesion
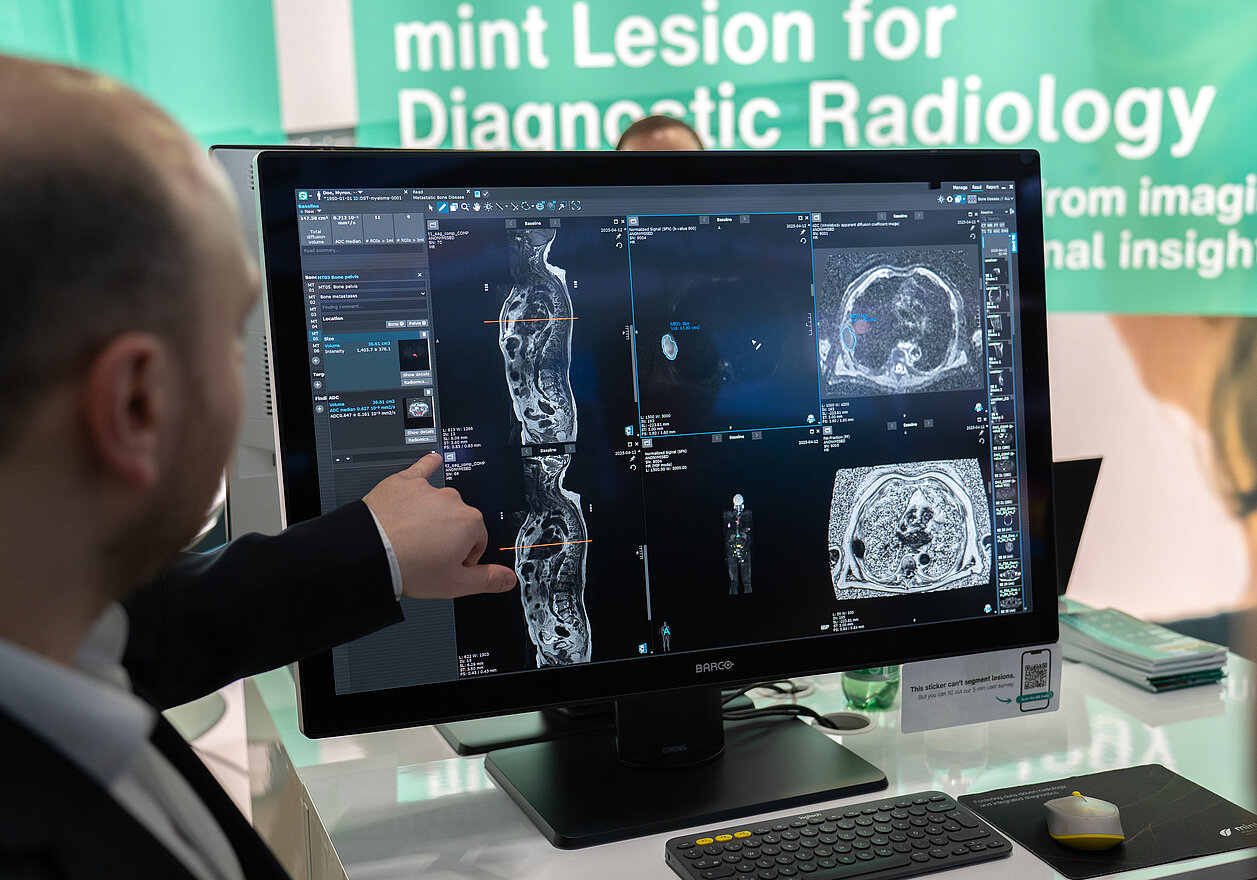
To address these workflow constraints, radiologists can utilize an AI algorithm* integrated into mint Lesion for the detection and segmentation of focal bone lesions. Developed in collaboration with The Royal Marsden NHS Foundation Trust and The Institute of Cancer Research, London, the software serves as a supportive framework that assists the radiologist in three primary areas:
- Automated Regional Analysis: Radiologists can analyze the disease burden within specific areas, as mint Lesion divides the skeleton into distinct body regions (such as the spine, pelvis, and long bones).
- Quantitative Disease Measurement: Radiologists obtain the total disease volume across the skeleton. They also gain access to disease volume and Apparent Diffusion Coefficient (ADC) values as a surrogate metric for cellularity, calculated on a per-region basis. To further evaluate the presence of active disease in the bone marrow, radiologists can cross-reference their DWI findings with automatically calculated fat fraction maps.
- Longitudinal Tracking: Radiologists can track active disease over time by utilizing mint Lesion’s side-by-side comparisons of serial scans, visual charts, and numerical evaluations.
Practical Outcomes for Clinical Workflows
The implementation of these tools aims to shift the reading process from qualitative estimation to data-supported reporting. This approach provides three primary outcomes:
- Practical Quantification: Supported by automated structural measurements, radiologists can execute full-skeleton quantification efficiently, overcoming the time barriers traditionally associated with manual analysis.
- Objective Metrics: mint Lesion provides reproducible metrics for risk stratification, moving away from subjective visual estimates like "looks slightly better" to concrete numerical changes.
- Data for Decision Support: Radiologists can provide oncologists and patients with objective, numerical data to back up their interpretations, offering a clearer baseline for treatment decisions.
*based on research funded by the National Institute for Health and Care Research (NIHR)
Interested in learning how AI-supported structured reporting can support your clinical workflows? Get in touch with our team to learn more about mint Lesion.