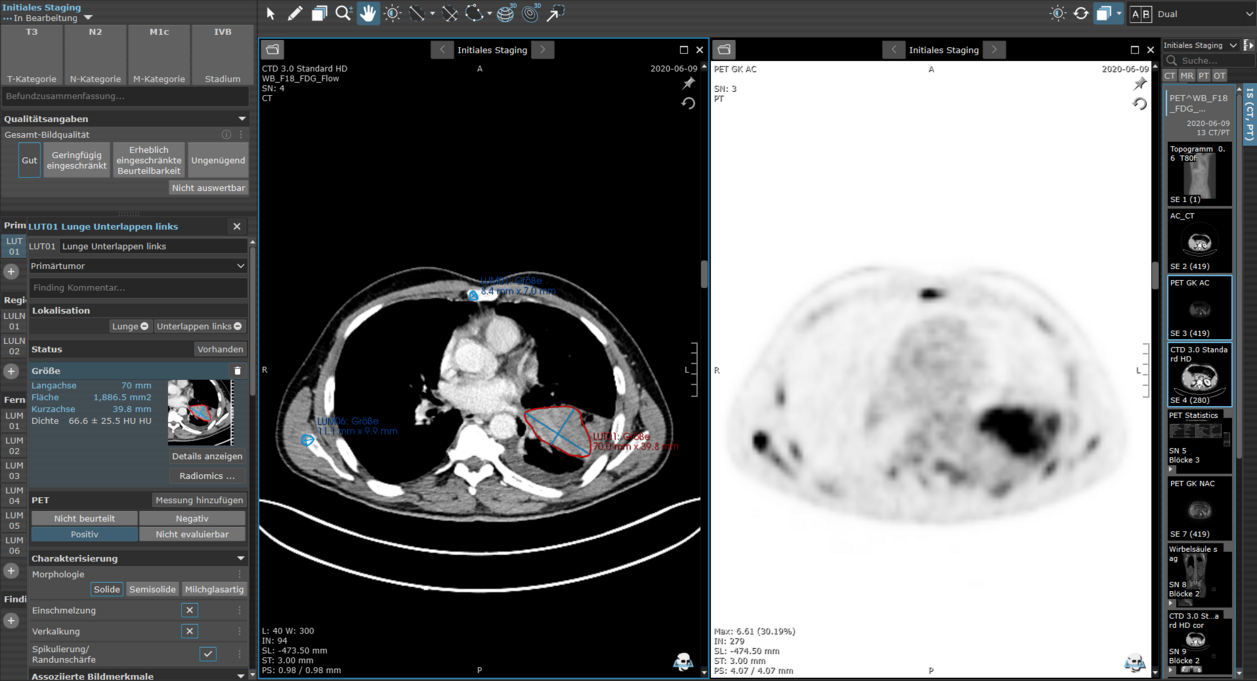
In this multi-center collaboration, thoracic radiology experts developed and evaluated a software-assisted structured reporting (SR) framework for staging non-small cell lung cancer (NSCLC). The study assessed the framework’s effectiveness in enhancing the accuracy of TNM classification and explored institutional standards, preferences, and perceptions of SR among participating institutions.
The software-assisted SR, featuring semi-automated TNM classification of NSCLC in mint Lesion significantly improved classification accuracy compared to traditional free-text reporting (FTR) (p = 0.01). Radiologists using SR were 1.707 times more likely to correctly classify TNM status. The most frequent classification errors were related to T-stage assessment, particularly in tumor size measurement and evaluation of local infiltration. Errors in N- and M-stage classification often resulted from incorrect lesion localization.
"The results of this proof-of-concept study suggest a valuable impact of software-assisted SR on TNM accuracy in NSCLC staging," the authors note.
The tool proved highly reliable, consistently generating accurate TNM classifications based on radiologists’ inputs. This improvement in classification accuracy led to a reduction in both upstaging and downstaging errors.
In a pre- and post-validation survey, participating radiologists expressed generally positive attitudes toward SR for NSCLC staging, rating its potential impact favorably across multiple categories. They acknowledged its value in improving report quality, completeness, and interdisciplinary communication. Positive perceptions of SR increased following hands-on use, indicating that practical experience may enhance its perceived benefits. Additionally, software-assisted SR improved radiologists' understanding of TNM criteria, suggesting these tools may serve as valuable educational aids.
The study also highlights SR's potential to support secondary data use and research. The standardized format of SR can facilitate the creation of large-scale registries, contributing to a deeper understanding of lung cancer trends and treatment outcomes.
The findings underscore the importance of addressing implementation challenges, such as digital infrastructure limitations, to fully realize SR’s potential in routine clinical practice.
This study was conducted as a collaborative effort within the Bavarian Oncologic Radiology Network (BORN) of the Bavarian Cancer Research Center (BZKF). Participating centers included radiology departments from LMU Hospital Munich, Technical University Munich, University Hospital Erlangen, University Hospital Regensburg, University Hospital Würzburg, and University Hospital Augsburg.
Read the original publication here.
Heimer, M.M., Dikhtyar, Y., Hoppe, B.F. et al. 2024. ”Software-assisted structured reporting and semi-automated TNM classification for NSCLC staging in a multicenter proof of concept study.” Insights Imaging.