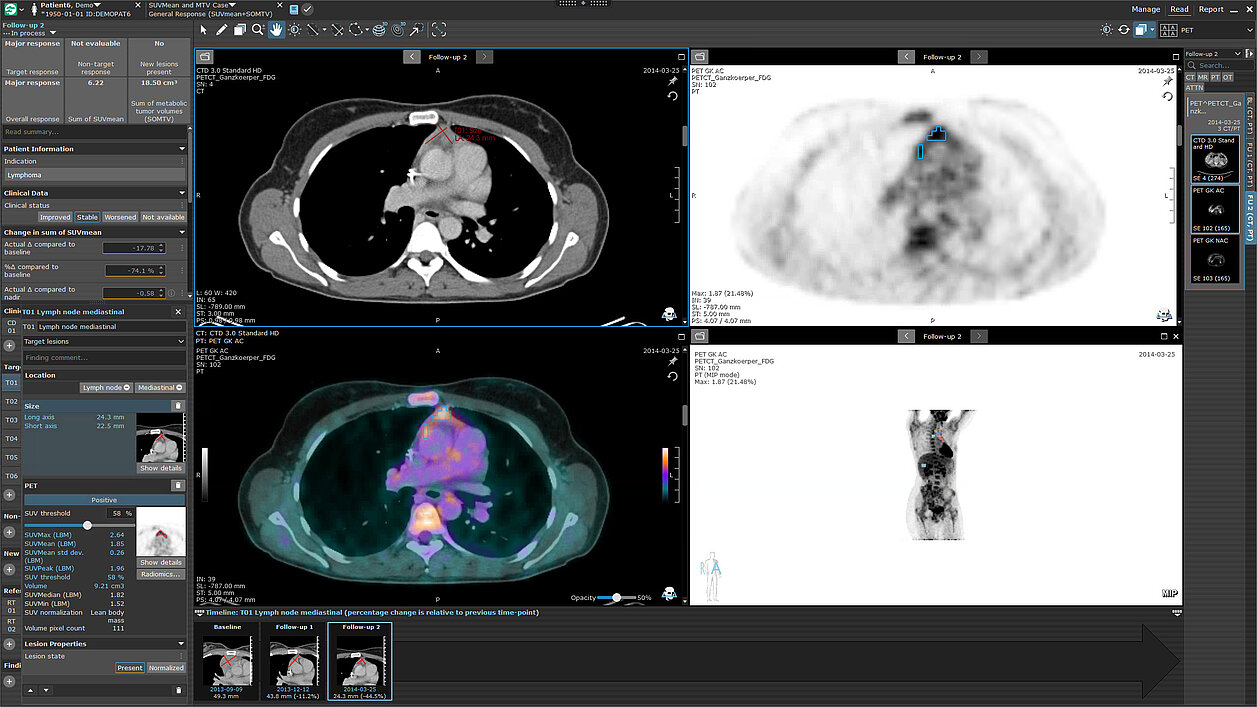
mint Lesion offers customizable reading templates for therapy response assessment in clinical care and research. Adapt parameters, define lesion categories, and evaluate responses. Track key measurements like diameter, volume, SUV, and ADC, ensuring compliance with required minimum sizes. Automate sum calculations for response determination and integrate clinical data such as ctDNA or PSA values. Document multiple lesions, compare absolute and relative changes over time, and visualize trends with interactive charts. Generate comprehensive reports with tables and graphs for a clear longitudinal overview.
Key features:
- Easily customizable parameters
- Questions for manual response evaluation
- Lesion categories (e.g., target lesions, non-target lesions)
- System-derived lesion states (e.g., normalized for lymph nodes)
- Key measurement values (e.g., longest diameter, short axis, volume, SUV, ADC, etc.), including required minimum size for each lesion category
- Sum of measurements for response determination (e.g., sum of diameter or sum of volume)
- Clinical data (e.g., ctDNA or PSA values)
- Ability to document the count of multiple (non-target and new) lesions
- Automatic calculations of absolute and relative differences to baseline, previous timepoint and nadir
- Interactive charts displaying all relevant values over time, including clinical data, with dynamic filtering options
- Comprehensive reports including tables and graphs for a fast and clear longitudinal overview