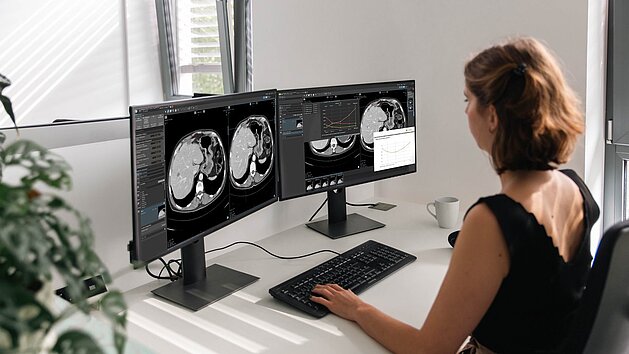
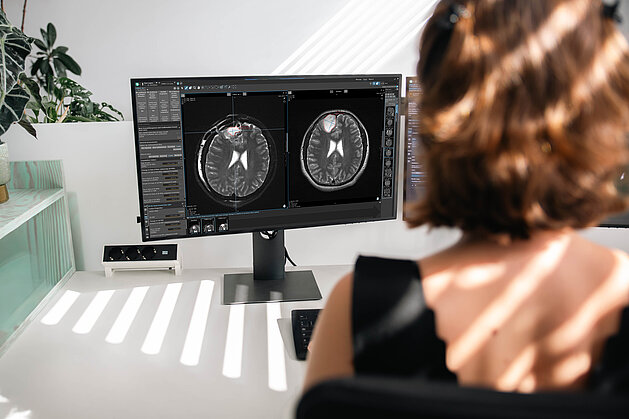
Requests for radiological clinical trial evaluations frequently rely on fragmented methods such as emails, phone calls, and spreadsheets. This decentralized approach often leads to communication delays, miscommunication, and challenges in maintaining clear traceability.
The mint Lesion web-based Clinical Trials Request Portal provides a centralized platform accessible from any department or workstation to enhance this process. By consolidating trial communications and workflows, the system assists in:
- Streamlining Communication: Connects radiology and trial teams directly to reduce administrative overhead.
- Full Traceability: Maintains a digital audit trail for all read requests, queries, and report approvals.
- Workflow Efficiency: Accelerates turnaround times for trial reads by organizing tasks in a unified interface.
- Protocol Compliance: Ensures structured, protocol-compliant workflows to support data quality.
To further support site operations, the portal includes specialized tools for:
- Patient and Trial Management: Manage trial data with integrated visit planning.
- Task Delegation: Assign roles and tasks to team members, including CRCs, radiologists, and PIs.
- Automated Query Management: Tools to create, receive, and resolve queries within the system to track communication history.
- Real-time Notifications: Automated email and chat alerts for updates and new requests.
- Web-Based PI Sign-Off: Digital interfaces for faster approvals and immediate documentation.
By integrating these functionalities into a single web-based environment, research sites can achieve a more organized approach to trial administration. This centralized framework supports consistent data management and provides clear visibility across the entire evaluation lifecycle.