"It's a paradigm shift." This is how Prof. Ulf Teichgräber, Director of the Institute of Diagnostic and Interventional Radiology at the University Hospital Jena, describes the improvement in communication with sponsors and CROs through the use of mint Lesion™. Together, Ms. Laura Graziani, study coordinator, Ms. Elisabeth Lammers, study assistant/MTRA, and Prof. Teichgräber describe their journey towards a successful clinical trial center that can now manage up to 40 clinical trials simultaneously.
Where results previously had to be painstakingly entered into Excel spreadsheets, mint Lesion™ has taken over the evaluation of all findings since its introduction in 2015, thus increasing the objectivity and validity of the study results.

Workflow optimization, increased efficiency and reduced errors in clinical trials
Related Resources
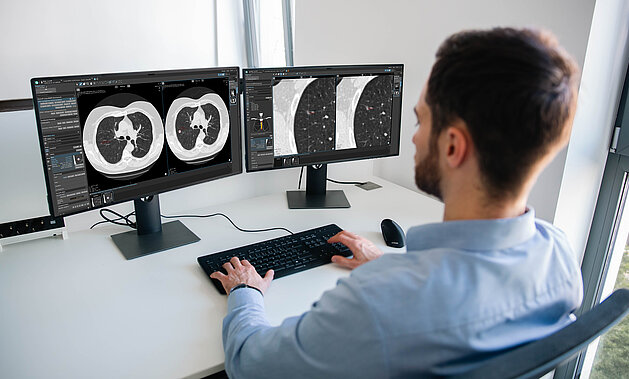
Lung Cancer Screening in Germany: How mint Lesion Supports Radiologists in Clinical Practice
A New Era of Early Detection — and New Challenges
With the introduction of the nationwide lung cancer screening program starting in 2026,…
Read more

From Theory to Practice: Hands-On RECIST Training at ECR 2026
This year, Mint Medical contributed to multiple program highlights at the ECR 2026 - bringing together innovation, education, and real-world clinical…
Read more
Webinar Series: Lung Cancer Screening in Germany - From Evidence to Implementation
Lung cancer remains one of the leading causes of cancer-related mortality worldwide. In Germany, the introduction of a national lung cancer screening…
Read more