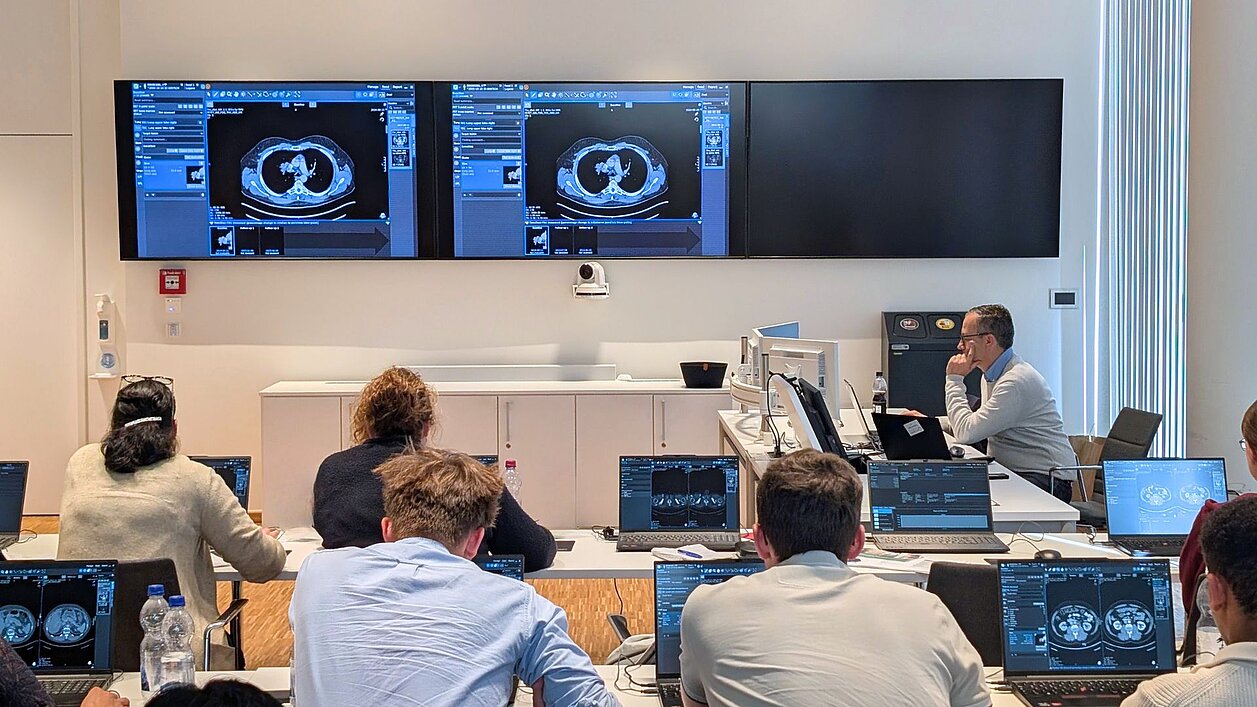
Successful “RECIST and Beyond” Workshop in Cologne: Advancing Precision in Oncologic Imaging
This central question shaped this year’s “RECIST and Beyond” workshop at the Radiological Center for Integrated Oncology (radCIO) at University Hospital Cologne. Over two intensive days in late September 2025, participants received exactly the practical, hands-on insights needed for their daily clinical routine.
Radiologists, oncologists, and specialists from related fields worked in an interactive hands-on format, performing their assessments directly in mint Lesion. This allowed them to immediately apply what they learned – structured, standardized, and fully traceable.

Day 1: Response Monitoring in Modern Oncology
The program focused on RECIST 1.1, iRECIST, mRECIST, LI-RADS Treatment Response, and the Lugano Classification. Participants learned how to reliably evaluate tumor response and progression, and how standardized reporting workflows enhance consistency and clarity in clinical decision-making.
Day 2: Liver and Biliary Tract Diagnostics with a Focus on LI-RADS
Saturday was dedicated to the classification, interpretation, and treatment response assessment of HCC as well as other hepatic and biliary tumors – highlighting the correct application of LI-RADS.

Learning in a Personalized Setting
With a deliberately limited number of participants, the workshop created an ideal environment for exchange, discussion, and individual feedback.
Prof. Dr. Thorsten Persigehl, scientific dorector of the event, summarized it perfectly:
“Through the independent hands-on training at the laptops, every participant will take something valuable back into their clinical routine reporting.”
We are delighted that we were able to support this workshop with mint Lesion, enabling participants to experience firsthand how structured reporting and decision support can increase confidence in everyday oncologic imaging.
More workshops are already in planning.
Follow us on LinkedIn to stay informed.
Related Resources

From Theory to Practice: Hands-On RECIST Training at ECR 2026
Webinar Series: Lung Cancer Screening in Germany - From Evidence to Implementation
