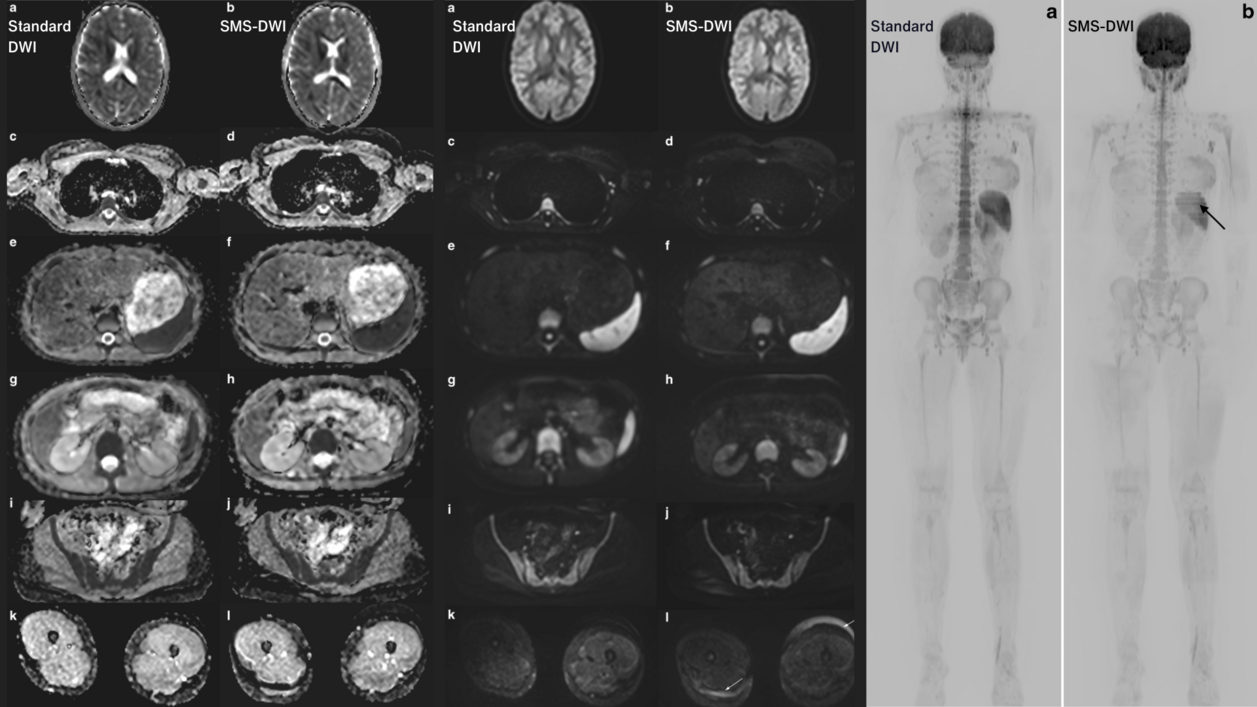
Researchers at the University Hospital Jena analyzed the response rate and safety of superselective drug-eluting beads transarterial chemoembolization (DEB-TACE) using doxorubicin-loaded 40-μm sized microspheres in patients with hepatocellular carcinoma (HCC) in a recent retrospective study [1].
Conventional TACE with lipiodol emulsion is still considered the current gold standard treatment for HCC. However, the chemotherapeutic agent is released into the bloodstream in an uncontrolled way. In contrast, DEB are permanent embolisates that hold doxorubicin in an ionic bond in the tumor. Several studies have shown the superiority of DEB-TACE using small- and medium-sized beads over conventional TACE: It leads to a higher tumor response and has less side effects. In addition, it was shown that smaller-sized microspheres allow for more homogeneous intratumoral agent distribution.
This study examined the therapeutic response and procedure-related complications after DEB-TACE using extremely small 40-μm microspheres. Furthermore, tumor characteristics such as longitudinal axis, volume, and texture parameters were investigated.
141 TACE interventions were performed in 83 patients, with a total of 282 follow-up examinations (255 CT and 27 MRI scans). The image evaluation was conducted using mint Lesion™ and was based on two different mRECIST assessments: First, a target-based evaluation of treated lesions only was performed (target response analysis). A second analysis was performed including all HCC lesions (overall response analysis).
In the target response analysis, the response rate (Complete Response/CR or Partial Response/PR) was 63.1% and the disease control rate (CR, PR or Stable Disease/SD) was 95.7%. The histogram analysis showed changes in values before and after each treatment cycle for entropy, uniformity, mean value of positive pixels (MPP), uniformity of distribution of positive pixels (UPP), minimal and maximal density (p<0.03 for all parameters).
The study is limited by its retrospective character and its monocentric implementation. Furthermore, since no comparison group was included (e. g. group of patients treated with conventional TACE or DEB-TACE with different particle sizes), “it cannot be claimed with certainty that the study outcomes are specific to the use of 40-μm microspheres,” the researchers explained. Therefore, they recommend further research to compare c-TACE with 40-μm DEB-TACE.
“This retrospective analysis supports the hypothesis that a high response rate in the treatment of HCC can be achieved using small-sized microspheres, which allow a deeper penetration of the nourishing tumor vessel,” the authors concluded. “This technique also minimizes the undesirable side effects of a false embolization.”
[1] Albrecht, K.C., Aschenbach, R., Diamantis, I. et al. Response rate and safety in patients with hepatocellular carcinoma treated with transarterial chemoembolization using 40-µm doxorubicin-eluting microspheres. J Cancer Res Clin Oncol (2020). https://doi.org/10.1007/s00432-020-03370-z