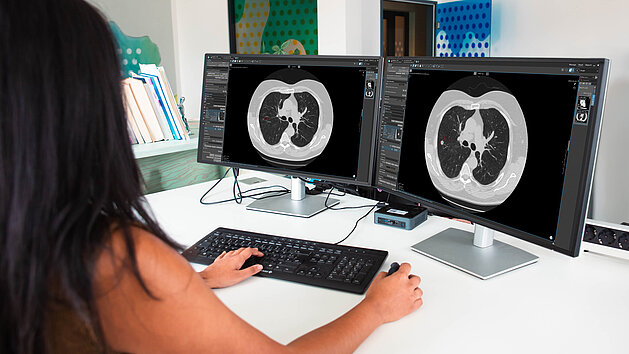
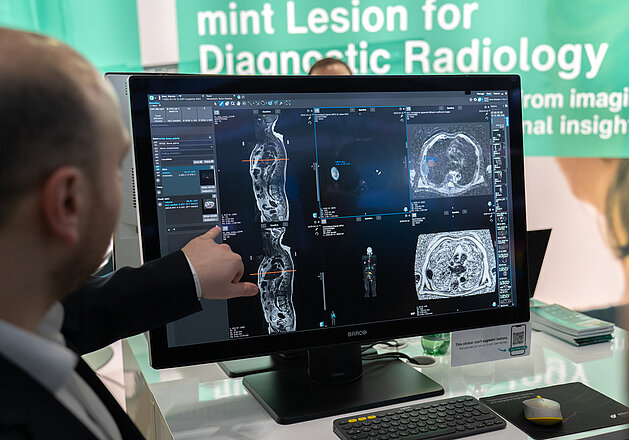
mint Lesion's Research Suite is an advanced imaging research platform designed to support academic teams in streamlining and scaling their imaging studies across single or multiple centers. Built to enhance research autonomy, the platform enables investigators to independently design, configure, and manage imaging studies with greater efficiency and consistency.
At its core, the Research Suite features our Template Designer that allows researchers to develop customized reporting templates tailored to their study objectives. Pre-configured workflows and defined user roles simplify study setup and coordination, supporting smoother operations across teams and sites.
Independent Study Configuration and Deployment
• Streamlined Study Setup
Researchers can configure their own studies using the Template Designer and pre-built workflows. The platform reduces dependency on IT teams by providing direct access to study configuration tools.
• Reduced Administrative Dependencies
The self-service approach allows researchers to manage study templates and configurations independently, minimizing wait times typically associated with IT support requests and administrative approvals.
• Integrated Testing Environment
Templates can be tested immediately after creation, allowing researchers to identify and address configuration issues before study deployment.
• Template Reusability
Created templates can be adapted and reused across different studies, supporting efficient scaling of research methodologies and reducing repetitive configuration work.
Multi-Center Research Coordination
• Multi-Center Deployment Capability
Local project configurations can be deployed across multiple research sites, supporting coordinated studies while maintaining site-specific requirements and protocols.
• Real-Time Study Oversight
Monitor global progress and data quality from a central dashboard, providing comprehensive visibility across all participating sites and enabling proactive study management.
• Automated De-Identification
The platform de-identifies patient data to support compliance with data-protection requirements while enabling centralized analytics.
Integrated Data Management and Analytics
• Integration of Imaging and EDC
The Research Suite combines imaging analysis with electronic data collection in a unified environment, eliminating the need to manage separate systems for measurements and data capture.
• Radiomics and Imaging Biomarker Extraction Built In
Extract quantitative imaging features and biomarkers directly within the platform - no extra tools or scripting required. Analytical capabilities are embedded into the standard workflow, making sophisticated imaging analysis accessible to the research teams.
• Instant Analytics
Visualize study data without programming or external tools. Built-in analytical functions and dashboards provide immediate insights into study metrics, quality indicators, and research outcomes throughout the study lifecycle.
Standardized Yet Flexible Framework
• Standardized Workflow Foundation
Pre-configured workflows and user roles provide consistent starting points for study setup, while maintaining flexibility for customization based on specific research requirements.
The Research Suite addresses common operational challenges in healthcare imaging research by providing researchers with direct control over study configuration and deployment processes.
The RACOON-SAGA project, conducted within the Network University Medicine (NUM), is one example of a study using the Research Suite. In this project, researchers aimed to improve the pretreatment characterization of soft tissue sarcomas by integrating imaging and clinical data.
You can contact us here if you'd like to know more.