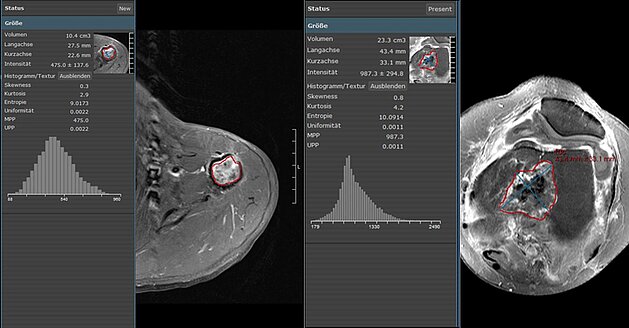
Mint Medical and Agfa HealthCare announce seamless integration of mint Lesion and ORBIS RIS at RSNA 2018
Heidelberg, Bonn, GER - At this year’s Radiological Society of North America (RSNA) Annual Meeting 2018, Agfa HealthCare and Mint Medical will…
Read more